Polyurethanes are widely used in biomedical applications such as artificial skin, hospital bedding, dialysis tubes, pacemaker components, catheters, and surgical coatings. The biocompatibility, mechanical properties, and low cost are major factors to the success of polyurethanes in the medical field.
The development of implants usually requires a high content of biobased components, because the body rejects them less. In the case of polyurethanes, the biocomponent can vary from 30 to 70%, which creates a broader scope for applications in such areas (2). The biobased polyurethanes are increasing their market share and are expected to reach about $42 million by 2022, which is a minuscule percentage of the overall polyurethane market (less than 0.1%). Nonetheless, it is a promising area, and intensive research is ongoing concerning the use of more biobased materials in polyurethanes. Improvement is needed in the properties of biobased polyurethanes to match the existing requirements, in order to scale up investment.
Biobased crystalline polyurethane was synthesized via a reaction of PCL, HMDI,and water that played the role of a chain extender (33). Degradation tests were performed to study the stability of biopolyurethane in simulated body fluids, such as phosphate-buffered saline solution. The changes
in thermal, mechanical, and physical properties were analyzed and compared to the equivalent
polyurethane obtained through using ethylene glycol as a chain extender instead of water. The results demonstrated that the polyurethane obtained using water as a chain extender presented better properties over time compared with its petrochemical equivalent. This not only greatly decreases
the cost of the process, but it also provides a facile route to obtaining value-added medical materials that are suitable for joint endoprostheses (33). This was followed by another approach based on this concept, which synthesized a biopolyurethane urea by utilizing rapeseed oil–based polyol, PCL,HMDI, and water as a chain extender (6). To increase the surface area, sodium chlorine was used to improve the porosity of the prepared polymers. The synthesized polymer was used as a scaffold due to its porous structure to induce cell growth of the bone tissue. With similar results compared
to the previous example, the polyurethane that was exposed to simulated body fluid presented high stability, providing a viable option for scaffold applications. Polyurethane ionomers are another interesting class of polymers used for biomedical applications, as a result of their biocompatibility and proper interaction with the body environment. Polyurethane ionomers can be used as tube components for pacemakers and hemodialysis (34, 35).
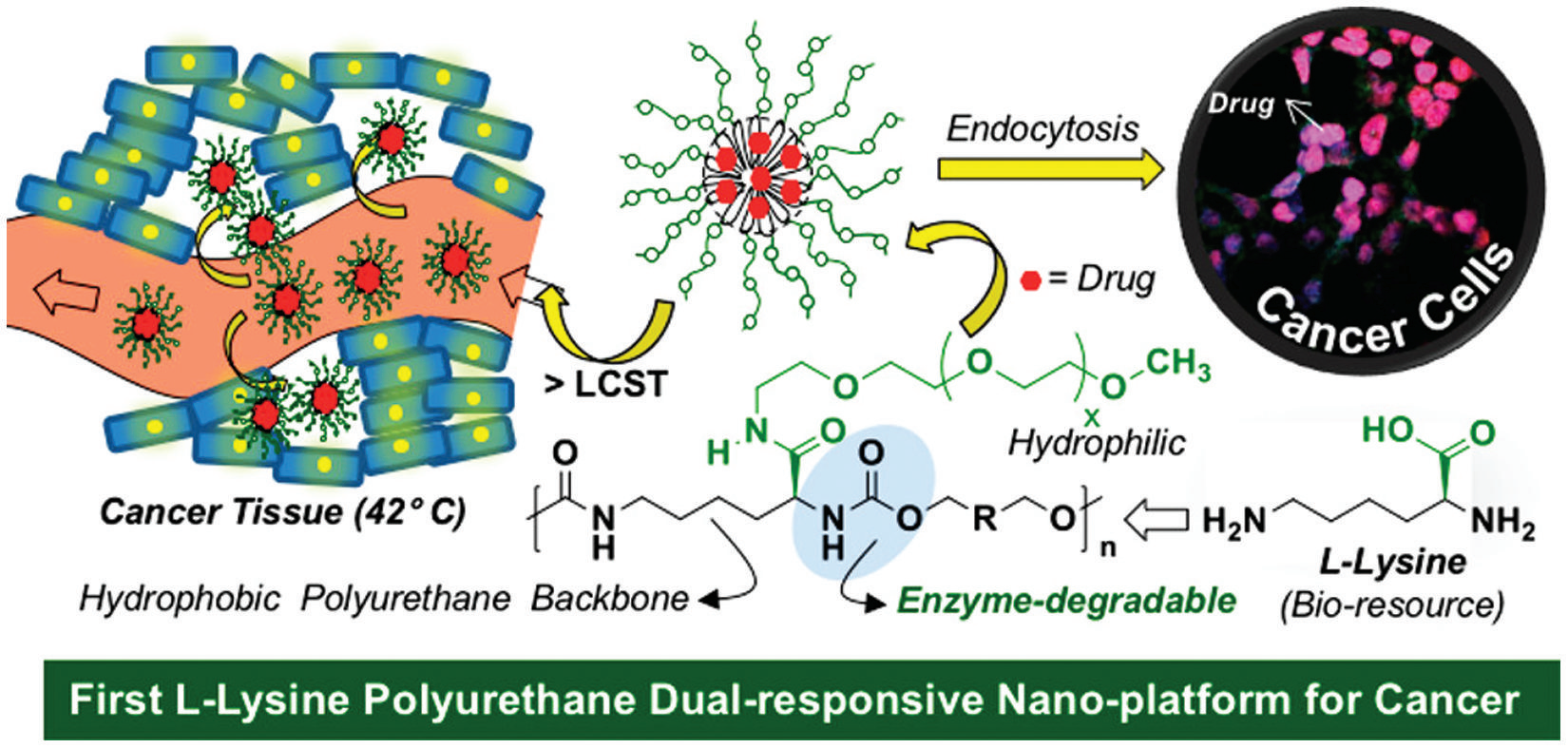
The development of an effective drug delivery system is an important research area that is currently focused on finding ways to tackle down cancer. An amphiphilic nanoparticle of polyurethane based on L-lysine was prepared for drug delivery applications (36). This nanocarrier
was effectively loaded with doxorubicin, which is an effective drug treatment for cancer cells (Figure 16). The hydrophobic segments of the polyurethane interacted with the drug, and the hydrophilic segments interacted with the cells. This system created a core-shell structure through a self-assembly
mechanism and was able to efficiently deliver drugs via two routes. First, the thermal response of the nanoparticle acted as a trigger in releasing the drug at the cancer cell’s temperature (~41–43 °C), which is an extracellular response. Second, the aliphatic segments of the polyurethane suffered
enzymatic biodegradation by the action of lysosomes,allowing doxorubicin to be released inside the cancer cell; this is an intracellular response. More than 90% of the breast cancer cells were killed, while low cytotoxicity was maintained for healthy cells.
Figure 16. Overall scheme for the drug delivery system based on an amphiphilic polyurethane nanoparticle
to target cancer cells. Reproduced with permission from reference (36). Copyright 2019 American Chemical
Society.
Declaration:The article is quoted from Introduction to Polyurethane Chemistry Felipe M. de Souza, 1 Pawan K. Kahol, 2 and Ram K.Gupta *,1 . Only for communication and learning, do not do other commercial purposes, does not represent the company’s views and opinions, if you need to reprint, please contact the original author, if there is infringement, please contact us immediately to do delete processing.
Post time: Nov-04-2022